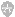包含体蛋白的纯化
2010-05-26 22:59:36 来源:不详 评论: 点击:
Buffer A
Buffer B
50 mM tris-HCl, pH 8.0
20 mM Na2HPO4, pH 7.2
5 mM EDTA
20 mM NaCl
| Buffer A |
|
Buffer B |
|
|
|
| 50 mM tris-HCl, pH 8.0 |
|
20 mM Na2HPO4, pH 7.2 |
| 5 mM EDTA |
|
20 mM NaCl |
| 10 mM NaCl |
|
5 mM EDTA |
|
|
25% (w/v) sucrose |
- Weigh the centrifuge bottle (w/cap) before adding culture media. You will need to know how many grams of cells you have before you start lysis.
- Spin down cells from large culture at 6000 rpm for 20 min. If you have a lot of media you can do multiple spins pouring out the supernatant after each spin.
- When spinning is complete weigh the flask (w/cap) and the cell pellet. Subtract the total weight from the empty flask weight to determine how many grams of cells you have.
- Add 3 mL of buffer A to the flask for each gram of cells and fully resuspend the cell pellet.
- Transfer the suspended pellet to 50 mL conical centrifuge tubes.
- Add enough 100 mM PMSF to make a 1 mM final solution (10ul 100 mM PMSF/ml of solution).
- Add 16 ul of 50mg/ml lysozyme per gram of cells and mix.
- Place in 37oC water bath until solution becomes viscous.
- Sonicate the solution to chop up the DNA and reduce the viscosity.
- Spin solution at 18,000 rpm for 30 min. Save supernatant.
- Add 3 mL of buffer B to the tube for each gram of cells and fully resuspend the cell pellet.
- Add the same amount of 100 mM PMSF as you did in step 6 and 10 ul Triton X-100 per ml of solution and resuspend the pellet.
- Spin at the highest speed possible (~20,000 rpm) for 20 min. Save supernatant. The pellet remaining will be the inclusion bodies.
- Add 20 mL of 8 mM urea (add DTT to 8 mM urea if purifying proteins with cysteine residues) and dissolve the pellet. Heating in a bath (37-50oC) may facilitate the pellet dissolving.
- Repeat step 14 until no more of the pellet dissolves.
- Dialyze 8 M urea solutions in 4 L of a 50 mM tris-HCl buffer solution at pH 8.5 using 3.5 kD molecular weight cutoff dialysis tubing for 2 days. Repeat, dumping out buffer and replacing with fresh buffer.
- Empty dialysis tubing into 50 mL conical centrifuge tubes and spin at 20,000 rpm for 30 min. Freeze supernant until HPLC purification.
- Clean up and wash all centrifuge tubes and flasks that you used.
Next step is to HPLC purify the protein. Before this step the solutions MUST be filtered through a 0.2u or 0.45u filter.