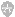This technique involves first laying down a thin layer of small matrix crystals on the sample stage. A droplet containing the analyte is placed on top of the crystalline deposit. The solvent containing the analyte is carefully chosen so that it will re dissolve part of the matrix layer. The resulting matrix/analyte solution dries, depositing the matrix on the seed sites provided by the undissolved portion of the original layer.
Two solutions must be prepared (A & B). The solvent for Solution A is acetone containing 1 - 2% water. Matrix is added to form a nearly saturated solution. A droplet (0.5 microliters) of solution A is placed on the sample stage and allowed to spread ou t and dry. The deposit formed should be uniform. The volume of solution A used and the matrix concentration depend on the geometry of the sample stage: the reader is encouraged to experiment with different conditions to suit his needs [24].
Solution B contains the peptide/protein of interest at a concentration of approximately 1 micromolar in an aqueous solution that contains 20 - 30% organic solvent. Place approximately 0.5 microliters of Solution B onto the surface prepared from Solution A and allow it to dry at room temperature. When the solution is completely dry, the deposit can be washed by placing a drop of pure water (or 0.1% aqueous TFA) onto the deposit for a 2 - 10 seconds and then removing excess water.