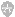First, prepare two solutions containing the matrix at room temperature, referred to below as Solution A and Solution B. Handle these solutions carefully: it is important to minimize transfer of undissolved crystalline solid from one step to the next. Prepare the solutions fresh each day.
Solution A contains the matrix alone. Prepare it using 0.3 ml of acetonitrile or any appropriate solvent mixture. Saturate the solvent with matrix in a 0.5 ml Eppendorf tube and swirl for at least one minute with a vortex mixer. Spin the tube in a benc h-top centrifuge to deposit undissolved matrix particles on the bottom of the tube. Remove the supernatant to another tube.
Prepare Solution B by saturating 0.3 ml of the selected solvent mixture with matrix and then remove excess matrix particles by centrifugation. Mix an aliquot of this matrix-saturated solution with a protein-containing solution to produce a final protein concentration of approximately 1 micromolar. It is important to remember that the solvent produced by mixing the protein-containing solution and the matrix-containing solution must keep both the matrix and the protein in solution. Precipitating either s olute leads to poor mass spectra.
To make the sample deposit, first place 0.5 - 1 microliters of Solution A on the mass spectrometer"s sample stage and allow it to dry. Place a clean glass slide onto the deposit and press down on it with an elastic rod, such as a pencil eraser. Twist th e glass slide several times to smear the deposit onto the surface and then remove the slide. Brush the crushed matrix with a tissue to remove any loose matrix particles. The layer of crushed matrix serves as the "activated" seeding site for the drying d roplet (see below). Alternatively, an activated layer can be made by electrospraying Solution A onto the sample stage, being careful that the sprayed layer does not become wet during the spray process. Electrospraying is a bit more difficult than simply crushing crystals, but the results are good and may be worth the trouble for particularly contaminated samples.
Apply 0.5 - 1 microliters of Solution B to the spot bearing the smeared matrix material. An opaque film forms over the substrate"s surface below the droplet within a few seconds, covering the metal. After about one minute, the probe tip can be immersed in room temperature water to remove involatile solvents and other contaminants. It is not necessary to let the droplet dry before washing: the film does not wash off easily. Remove excess water and allow it to dry before loading into the mass spectrome ter.
The aliquot of Solution B used to create the film must be free of particulate matter. Any particles of matrix present in this solution result in rapid nucleation of matrix crystals throughout the solution, interfering with the formation of the film. If Solution B is obviously cloudy, further centrifugation is necessary to clear the liquid.